Since the advent of cisplatin, the pioneering platinum-based chemotherapeutic approved in 1978, platinum compounds have become a cornerstone in oncology due to their broad spectrum of anti-tumor activity [1]. To date, platinum-based drugs have been widely used for the treatment of various cancers, such as lung cancer, ovarian cancer, and bladder cancer [2,3]. Despite their clinical success, drug resistance to platinum compounds has emerged as a significant challenge, necessitating the exploration of new therapeutic strategies [[4], [5], [6]].
Ruthenium complexes have demonstrated significant potential in cancer therapy due to their unique mechanisms of action [7,8]. Unlike platinum drugs that primarily damage nuclear DNA, Ru complexes exploit the hyperpolarized mitochondrial membrane potential of cancer cells to selectively accumulate in mitochondria, disrupting energy metabolism and directly triggering apoptosis [9]. For instance, ruthenium complexes can exert anticancer effects through photochemical decomposition, reduction, or photodynamic effects. The heavy atom effect of the central metal enhances spin-orbit coupling, significantly improving the efficacy of photodynamic therapy [10].Additionally, rational molecular design enables the construction of ruthenium complexes with intrinsic anticancer activity, reducing reliance on external stimuli and enhancing treatment precision [11,12]. However, current methods still face challenges in regulating drug activity and controlling degradation [13,14]. Traditional encapsulation techniques often struggle to achieve precise drug release, while recent advances in nanodelivery systems, although promising for targeted delivery and controlled release, encounter issues such as poor degradability or dependence on external light activation [[15], [16], [17]]. Therefore, the development of ruthenium-based anticancer agents that combine controlled release, biodegradability, and high therapeutic efficacy has become a focal point of research [18,19]. This study aims to explore novel strategies for optimizing the design and delivery of ruthenium complexes to advance their application in cancer therapy.
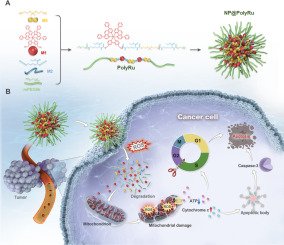
In this study, we have developed PolyRu, a main-chain Ru polymer that integrates ROS-responsive drug release, mitochondrial targeting, and immune activation into a single nanoplatform, offering a mechanistically distinct strategy to combat drug-resistant cancers. NP@PolyRu was prepared through the self-assembly of a novel amphiphilic polymer, PolyRu, which contains a Ru complex (Ru(II)-OH) and thioketal bonds in its main chain. The thioketal bonds in NP@PolyRu can be degraded by high concentrations of ROS after cancer cell uptake without having to go through exogenous stimulation, leading to the release of Ru(II)-OH. Ru(II)-OH can then selectively target cancer cell mitochondria to induce mitochondrial damage and impair energy metabolism, leading to cancer cell apoptosis. In a patient-derived xenograft (PDX) model of human lung cancer, NP@PolyRu effectively inhibited the progression of cisplatin-resistant tumors, marking a promising advancement in overcoming cancer drug resistance.