Among various electrochemical energy storage systems, aqueous zinc-ion batteries (ZIBs) with the merits of high safety, environmental friendliness and low cost have received extensive attention in recent years for their promising applications as grid-scale energy storage systems [1,2]. The relatively low redox potential (−0.76 V vs. standard hydrogen electrode) and large theoretical specific capacity (820 mAh/g) of zinc metal anode are conducive to improving the energy density of ZIBs, while the aqueous zinc-salt electrolytes featured by non-inflammability and high ionic conductivity endow ZIBs with both intrinsic safety and the potential of realizing fast charge/discharge rate [3]. On this basis, the ion storage behaviors of cathode materials play an important role in the electrochemical properties of ZIBs. Although divalent Zn2+ carries more charges than univalent cations and its storage in cathode host materials is promising to achieve prominent ion-storage capacity, the strong electrostatic interaction between Zn2+ and cathode host materials makes many univalent-ion host materials incapable of storing Zn2+, leading to the scarcity of high-performance cathode materials for Zn2+ storage to restrict the development of ZIBs [4].
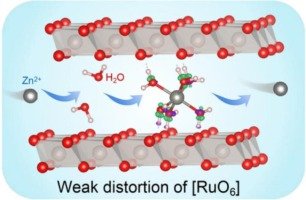
Transition metal oxides (TMOs) especially Mn-based and V-based TMOs have attracted broad attention in aqueous ZIBs, since their appropriate crystal structures (e.g., layered and tunnel-structured) and abundant redox active sites favor divalent Zn2+ diffusion and storage [1,5]. Nevertheless, Mn-based and V-based TMO cathode materials suffer from inferior cycling stability, which is associated with Zn2+ insertion-caused large lattice distortion and water molecule-induced dissolution in ZIBs. To prolong the cycling life of TMO cathodes, plentiful strategies such as ion/molecule pre-intercalation, micro-nano structure regulation and electrolyte engineering have been proposed. Specifically, pre-intercalated interlayer ions and molecules increase the interlayer spacing of TMO cathode materials, thereby weakening the electrostatic interaction between Zn2+ and the TMO hosts to reduce Zn2+ diffusion resistance and enhance the TMO structural durability during repeated Zn2+ insertion/desertion [6,7]. Regulating micro-nano structure is expected to improve the Zn2+-storage stability of TMO cathode materials by exposing more redox active sites on cathode-electrolyte interfaces to shorten Zn2+/electron diffusion distance [8]. In addition, electrolyte engineering, including solvation structure modulation to decrease water activity and electrolyte composition regulation to inhibit TMO dissolution, is also beneficial for optimizing the cycling performance of TMO cathodes [9]. Nonetheless, those approaches seldom fundamentally eliminate Zn2+ insertion-induced lattice distortions of TMO materials, resulting in suboptimal Zn2+-storage stability for the current TMO cathodes. To be specific, in [TMO6] octahedra-constructed compounds, certain electronic configurations will lead to a geometrical distortion in the crystal structure to lower the symmetry and energy of the system, which is referred to as the Jahn-Teller distortion [10]. The transition metal ions obviously showing this phenomenon are named Jahn-Teller active ions, whereas the opposite is Jahn-Teller inactive ions [11]. As for Jahn-Teller active TMO materials, Zn2+ storage results in the change of electronic configuration from a low-spin state to a high-spin state, inducing asymmetric lattice distortions in the octahedra-constructed compounds to generate lattice stresses and aggravate irreversible phase transition or structure collapse, which thereby accelerates the capacity fading of TMO cathode materials. Taking the Mn-based TMO cathode materials as an example, with the reduction of Mn4+ (low-spin t2g3eg0) to Mn3+ (high-spin t2g3eg1) during the Zn2+ storage process, the electron distribution of Mn3+ with only one of the doubly degenerate orbits being occupied (half-filled dz2 and empty dx2-y2) will induce large axial elongation of the [MnO6] octahedra to remove the orbital degeneracy and reduce the energy of the system, in which case severe lattice distortion and its caused structural instability are inevitable for Mn-based cathode materials [12,13]. Therefore, apart from the strong electrostatic interactions between Zn2+ and TMO hosts, Jahn-Teller effect-induced inherent lattice instability is also a crucial reason for the inferior cycling performance of TMO cathodes for ZIBs [14,15]. From this viewpoint, exploring TMO cathode materials with weak Jahn-Teller distortion is promising to achieve stable Zn2+ storage, whereas it has rarely been studied.
 O bonds during Zn2+ storage, and simultaneously, the interlayer water molecules not only dampen the charge transfer between inserted Zn2+ and the lattice oxygen to weaken the electrostatic interaction between them but also induce Zn2+ moving through low-energy-barrier integral rotation of octahedral [Zn(H2O)6]2+ cluster to decrease Zn2+ diffusion resistance in the nanochannels of the cathode material. As a result, the synergistic effect between low lattice distortion and interlayer water molecules enables long-term stable Zn2+ storage for the HRO·0.72H2O cathode material.
O bonds during Zn2+ storage, and simultaneously, the interlayer water molecules not only dampen the charge transfer between inserted Zn2+ and the lattice oxygen to weaken the electrostatic interaction between them but also induce Zn2+ moving through low-energy-barrier integral rotation of octahedral [Zn(H2O)6]2+ cluster to decrease Zn2+ diffusion resistance in the nanochannels of the cathode material. As a result, the synergistic effect between low lattice distortion and interlayer water molecules enables long-term stable Zn2+ storage for the HRO·0.72H2O cathode material.