Pop, F., Zigon, N. & Avarvari, N. Main-group-based electro- and photoactive chiral materials. Chem. Rev. 119, 8435–8478 (2019).
Hu, J., Ferger, M., Shi, Z. & Marder, T. B. Recent advances in asymmetric borylation by transition metal catalysis. Chem. Soc. Rev. 50, 13129–13188 (2021).
Di, J., Han, S. & Chen, P. Recent advances of boron-containing chiral luminescent materials. Chin. J. Chem. 43, 219–238 (2025).
Yang, K. & Song, Q. Tetracoordinate boron intermediates enable unconventional transformations. Acc. Chem. Res. 54, 2298–2312 (2021).
Staubitz, A., Robertson, A. P. M., Sloan, M. E. & Manners, I. Amine- and phosphine-borane adducts: new interest in old molecules. Chem. Rev. 110, 4023–4078 (2010).
Staubitz, A., Robertson, A. P. M. & Manners, I. Ammonia-borane and related compounds as dihydrogen sources. Chem. Rev. 110, 4079–4124 (2010).
Curran, D. P. et al. Synthesis and reactions of N-heterocyclic carbene boranes. Angew. Chem. Int. Ed. 50, 10294–10317 (2011).
Cuenca, A. B., Shishido, R., Ito, H. & Fernández, E. Transition-metal-free B–B and B–interelement reactions with organic molecules. Chem. Soc. Rev. 46, 415–430 (2017).
Hütter, R. et al. Stoffwechselprodukte von Mikroorganismen. 57. mitteilung. boromycin. Helv. Chim. Acta 50, 1533–1539 (1967).
Haefele, A. et al. Boron asymmetry in a BODIPY derivative. Org. Lett. 12, 1672–1675 (2010).
Hu, D., Huang, R. & Fang, Y. Recent advances in tetra-coordinate boron-based photoactive molecules for luminescent sensing, imaging, and anticounterfeiting. Precis. Chem. 3, 10–26 (2025).
Uraguchi, D. et al. A structurally robust chiral borate ion: molecular design, synthesis, and asymmetric catalysis. Angew. Chem. Int. Ed. 59, 11456–11461 (2020).
Xu, J. et al. Palladium-catalyzed atroposelective kinetic C−H olefination and allylation for the synthesis of C−B axial chirality. Angew. Chem. Int. Ed. 62, e202313388 (2023).
Matteson, D. S. -Halo boronic esters: intermediates for stereodirected synthesis. Chem. Rev. 89, 1535–1551 (1989).
Collins, B. S. L., Wilson, C. M., Myers, E. L. & Aggarwal, V. K. Asymmetric synthesis of secondary and tertiary boronic esters. Angew. Chem. Int. Ed. 56, 11700–11733 (2017).
Namirembe, S. & Morken, J. P. Reactions of organoboron compounds enabled by catalyst-promoted metalate shifts. Chem. Soc. Rev. 48, 3464–3474 (2019).
Sharma, H. A., Essman, J. Z. & Jacobsen, E. N. Enantioselective catalytic 1,2-boronate rearrangements. Science 374, 752–757 (2021).
Zhang, X., Gao, C. & Morken, J. P. Enantioselective construction of carbocyclic and heterocyclic tertiary boronic esters by conjunctive cross-coupling reaction. J. Am. Chem. Soc. 145, 16344–16349 (2023).
Jiang, X.-M. et al. Asymmetric synthesis of chiral 1,2-bis(boronic) esters featuring acyclic, non-adjacent 1,3-stereocenters. Angew. Chem. Int. Ed. 63, e202318441 (2024).
Guo, Y., Zu, B., Du Chen, C. & He, C. Boron-stereogenic compounds: synthetic developments and opportunities. Chin. J. Chem. 42, 2401–2411 (2024).
Zhang, X. et al. Phase-transfer-catalyst enabled enantioselective C–N coupling to access chiral boron-stereogenic BODIPYs. Nat. Commun. 16, 2735 (2025).
Schraff, S., Sun, Y. & Pammer, F. Tuning of electronic properties via labile N→B-coordination in conjugated organoboranes. J. Mater. Chem. C. 5, 1730–1741 (2017).
Mellerup, S. K. & Wang, S. Boron-based stimuli responsive materials. Chem. Soc. Rev. 48, 3537–3549 (2019).
Huang, X. & Ma, S. Allenation of terminal alkynes with aldehydes and ketones. Acc. Chem. Res. 52, 1301–1312 (2019).
Li, G., Huo, X., Jiang, X. & Zhang, W. Asymmetric synthesis of allylic compounds via hydrofunctionalisation and difunctionalisation of dienes, allenes, and alkynes. Chem. Soc. Rev. 49, 2060–2118 (2020).
Corpas, J., Mauleón, P., Arrayás, R. G. & Carretero, J. C. Transition-metal-catalyzed functionalization of alkynes with organoboron reagents: new trends, mechanistic insights, and applications. ACS Catal. 11, 7513–7551 (2021).
Liu, C.-X. et al. Rhodium-catalyzed asymmetric C−H functionalization reactions. Chem. Rev. 123, 10079–10134 (2023).
Shu, T. & Cossy, J. Asymmetric desymmetrization of alkene-, alkyne- and allene-tethered cyclohexadienones using transition metal catalysis. Chem. Soc. Rev. 50, 658–666 (2021).
Nájera, C., Foubelo, F., Sansano, J. M. & Yus, M. Enantioselective desymmetrization reactions in asymmetric catalysis. Tetrahedron 106, 132629–132756 (2022).
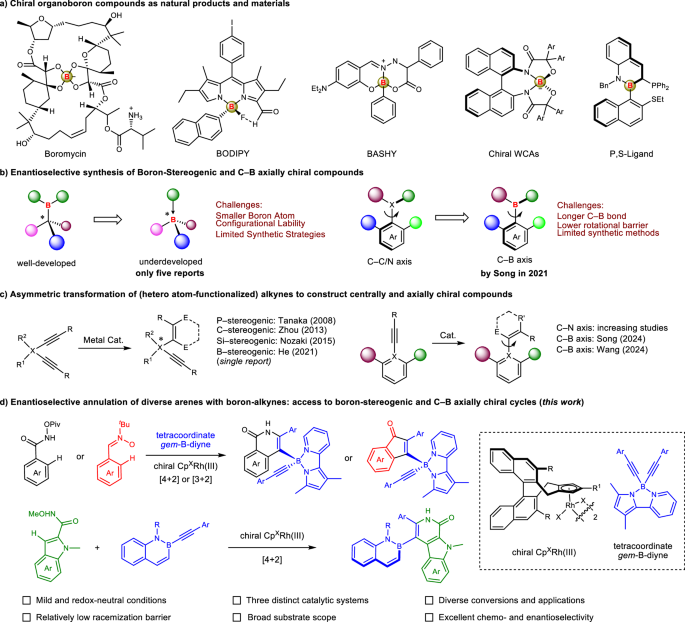
Nishida, G., Noguchi, K., Hirano, M. & Tanaka, K. Enantioselective synthesis of P-stereogenic alkynylphosphine oxides by Rh-catalyzed [2+2+2] cycloaddition. Angew. Chem. Int. Ed. 47, 3410–3413 (2008).
Zhou, F. et al. Asymmetric copper(I)-catalyzed azide alkyne cycloaddition to quaternary oxindoles. J. Am. Chem. Soc. 135, 10994–10997 (2013).
Shintani, R., Takagi, C., Ito, T., Naito, M. & Nozaki, K. Rhodium-catalyzed asymmetric synthesis of silicon-stereogenic dibenzosiloles by enantioselective [2+2+2] cycloaddition. Angew. Chem. Int. Ed. 54, 1616–1620 (2015).
Pierrot, D. & Marek, I. Synthesis of enantioenriched vicinal tertiary and quaternary carbon stereogenic centers within an acyclic chain. Angew. Chem. Int. Ed. 59, 36–49 (2020).
Ye, F., Xu, Z. & Xu, L.-W. The discovery of multifunctional chiral P ligands for the catalytic construction of quaternary carbon/silicon and multiple stereogenic centers. Acc. Chem. Res. 54, 452–470 (2021).
Xu, L.-W., Li, L., Lai, G.-Q. & Jiang, J.-X. The recent synthesis and application of silicon-stereogenic silanes: a renewed and significant challenge in asymmetric synthesis. Chem. Soc. Rev. 40, 1777–1790 (2011).
Ge, Y., Ke, J. & He, C. Catalytic asymmetric dehydrogenative Si-H/X-H coupling toward Si-stereogenic silanes. Acc. Chem. Res. 58, 375–398 (2025).
Zhao, J., Ge, Y. & He, C. Construction of silicon-stereogenic center via catalytic asymmetric Si-H/X-H dehydrogenative coupling. Chin. J. Org. Chem. 43, 3352–3366 (2023).
Harvey, J. S. & Gouverneur, V. Catalytic enantioselective synthesis of P-stereogenic compounds. Chem. Commun. 46, 7477–7485 (2010).
Liu, S., Li, Y., Wang, D., Wei, R. & Miao, Z. Research progress of asymmetric synthesis of optically active P-stereogenic organophosphoryl compounds by chiral induction. Chin. J. Org. Chem. 38, 341–349 (2018).
Zu, B., Guo, Y. & He, C. Catalytic enantioselective construction of chiroptical boron stereogenic compounds. J. Am. Chem. Soc. 143, 16302–16310 (2021).
Zhang, G. et al. Construction of boron-stereogenic compounds via enantioselective Cu-catalyzed desymmetric B–H bond insertion reaction. Nat. Commun. 13, 2624 (2022).
Zu, B., Guo, Y., Ren, L.-Q., Li, Y. & He, C. Catalytic enantioselective synthesis of boron-stereogenic BODIPYs. Nat. Synth. 2, 564–571 (2023).
Ren, L.-Q. et al. Modular enantioselective assembly of multi-substituted boron-stereogenic BODIPYs. Nat. Chem. 17, 83–91 (2025).
Zhan, B. et al. Catalytic asymmetric C–N cross-coupling towards boron-stereogenic 3-amino-BODIPYs. Nat. Commun. 16, 438 (2025).
Bringmann, G., Gulder, T., Gulder, T. A. M. & Breuning, M. Atroposelective total synthesis of axially chiral biaryl natural products. Chem. Rev. 111, 563–639 (2011).
Kumarasamy, E., Raghunathan, R., Sibi, M. P. & Sivaguru, J. Nonbiaryl and heterobiaryl atropisomers: molecular templates with promise for atropselective chemical transformations. Chem. Rev. 115, 11239–11300 (2015).
Cheng, J. K., Xiang, S. H. & Tan, B. Organocatalytic enantioselective synthesis of axially chiral molecules: development of strategies and skeletons. Acc. Chem. Res. 55, 2920–2937 (2022).
Basilaia, M., Chen, M. H., Secka, J. & Gustafson, J. L. Atropisomerism in the pharmaceutically relevant realm. Acc. Chem. Res. 55, 2904–2919 (2022).
Bai, X.-F., Cui, Y.-M., Cao, J. & Xu, L.-W. Atropisomers with axial and point chirality: synthesis and applications. Acc. Chem. Res. 55, 2545–2561 (2022).
Wencel-Delord, J., Panossian, A., Leroux, F. R. & Colobert, F. Recent advances and new concepts for the synthesis of axially stereoenriched biaryls. Chem. Soc. Rev. 44, 3418–3430 (2015).
Jin, L. et al. Atroposelective synthesis of axially chiral styrenes via an asymmetric C–H functionalization strategy. Chem. 6, 497–511 (2020).
Xiang, S.-H., Ding, W.-Y., Wang, Y.-B. & Tan, B. Catalytic atroposelective synthesis. Nat. Catal. 7, 483–498 (2024).
Mei, G.-J., Koay, W.-L., Guan, C.-Y. & Lu, Y. Atropisomers beyond the C–C axial chirality: advances in catalytic asymmetric synthesis. Chem 8, 1855–1893 (2022).
Kitagawa, O. Structural chemistry of C–N axially chiral compounds. J. Org. Chem. 89, 11089–11099 (2024).
Rodriguez-Salamanca, P., Fernández, R., Hornillos, V. & Lassaletta, J. M. Asymmetric synthesis of axially chiral C–N atropisomers. Chem. Eur. J. 28, e202104442 (2022).
Feng, J., Lu, C.-J. & Liu, R.-R. Catalytic asymmetric synthesis of atropisomers featuring an aza axis. Acc. Chem. Res. 56, 2537–2554 (2023).
Centonze, G., Portolani, C., Righi, P. & Bencivenni, G. Enantioselective strategies for synthesis of N-N atropisomers. Angew. Chem. Int. Ed. 62, e202303966 (2023).
Feng, J. & Liu, R.-R. Catalytic asymmetric synthesis of N−N biaryl atropisomers. Chem. Eur. J. 30, e202303165 (2024).
Mancinelli, M., Bencivenni, G., Pecorari, D. & Mazzanti, A. Stereochemistry and recent applications of axially chiral organic molecules. Eur. J. Org. Chem. 2020, 4070–4086 (2020).
Yang, K. et al. Construction of axially chiral arylborons via atroposelective Miyaura borylation. J. Am. Chem. Soc. 143, 10048–10053 (2021).
Wang, H. et al. Enantio- and regioselective [2+2+2] cycloaddition of BN-diynes for construction of C−B axial chirality. Chem. 10, 317–329 (2024).
Ping, Y., Shi, X., Lei, M. & Wang, J. Atroposelective construction of carbon-boron axial chirality through Rh-catalyzed [2+2+2] cycloaddition. ACS Catal. 14, 5064–5076 (2024).
Qiu, W. et al. Enantioselective construction of C−B axially chiral alkenylborons by nickel-catalyzed radical relayed reductive coupling. Nat. Commun. 15, 10432 (2024).
Yang, K. et al. Construction of C−B axial chirality via dynamic kinetic asymmetric cross-coupling mediated by tetracoordinate boron. Nat. Commun. 14, 4438 (2023).
Wang, F. et al. Rhodium(III)-catalyzed atroposelective synthesis of biaryls by C−H activation and intermolecular coupling with sterically hindered alkynes. Angew. Chem. Int. Ed. 59, 13288–13294 (2020).
Wang, F. et al. Rhodium-catalyzed C−H activation-based construction of axially and centrally chiral indenes through two discrete insertions. Angew. Chem. Int. Ed. 60, 16628–16633 (2021).
Mi, R. et al. O-Allylhydroxyamine: a bifunctional olefin for construction of axially and centrally chiral amino alcohols via asymmetric carboamidation. J. Am. Chem. Soc. 145, 8150–8162 (2023).
Wang, P. et al. Sigma-bond metathesis as an unusual asymmetric induction step in rhodium-catalyzed enantiodivergent synthesis of C−N axially chiral biaryls. J. Am. Chem. Soc. 145, 8417–8429 (2023).
Wang, Y. et al. Rhodium-catalyzed enantioselective and diastereodivergent access to diaxially chiral heterocycles. Nat. Commun. 14, 4661 (2023).
Zhu, X., Mi, R., Yin, J., Wang, F. & Li, X. Rhodium-catalyzed atroposelective access to trisubstituted olefins via C−H bond olefination of diverse arenes. Chem. Sci. 14, 7999–8005 (2023).
Zhu, X. et al. Rhodium-catalyzed annulative approach to N–N axially chiral biaryls via C–H activation and dynamic kinetic transformation. Chem. Sci. 14, 8564–8569 (2023).
Ye, B. & Cramer, N. A tunable class of chiral Cp ligands for enantioselective rhodium(III)-catalyzed C−H allylations of benzamides. J. Am. Chem. Soc. 135, 636–639 (2013).
Guimond, N., Gorelsky, S. I. & Fagnou, K. Rhodium(III)-catalyzed heterocycle synthesis using an internal oxidant: improved reactivity and mechanistic studies. J. Am. Chem. Soc. 133, 6449–6457 (2011).
Hopfl, H. The tetrahedral character of the boron atom newly defined – a useful tool to evaluate the N→B bond. J. Organomet. Chem. 581, 129–149 (1999).
Selby, J. et al. Chiroptical properties of indolenine squaraines with a stereogenic center at close proximity. J. Org. Chem. 85, 12227–12242 (2020).