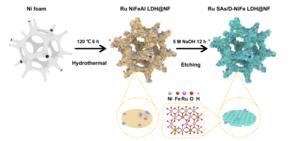
image:
Ru SAs were integrated into defective NiFe LDH (Ru SAs/D-NiFe LDH@NF). This was done on the nickel foam framework through a simple hydrothermal reaction followed by an etching process. The implanted Ru SAs synergistically optimized the local electronic structure by redistributing charge density surrounding adjacent Ni/Fe sites. It enhanced adsorption capacity for OER intermediates and promoted reaction kinetics.
Credit: Nano Research, Tsinghua University Press
The inefficiency of oxygen evolution reaction (OER) catalysts has long constrained the development of green energy devices, with traditional precious metal catalysts facing bottlenecks of high cost and poor stability. Recently, a team led by Dr. Hu from Chongqing University of Technology, in collaboration with Hainan University, developed a defect-type nickel-iron layered double hydroxide (LDH) catalyst anchored with ruthenium single atoms (Ru SAs/D-NiFe LDH@NF). This breakthrough successfully elevated the performance of zinc-air batteries to unprecedented levels. Published in Nano Research, this achievement offers a novel solution for highly efficient energy conversion technologies.
This material was synthesized via a two-step hydrothermal-etching process: first, a ruthenium nickel-iron-aluminum hydrotalcite precursor was grown on a nickel foam substrate; then, an alkaline solution selectively etched aluminum ions to form defect anchor sites, ultimately enabling precise anchoring of ruthenium single atoms (loading 2.4%).
Experiments demonstrate that this “defect engineering + single-atom synergy” strategy enables the catalyst to drive a current density of 50 mA cm-2 with only 206 mV overpotential in alkaline media, significantly lower than pristine NiFe LDH (311 mV) and commercial Ir/C (355 mV). Its exceptional activity stems from ruthenium atoms restructuring the electronic framework of defect sites—density functional theory calculations confirm that ruthenium’s high electronegativity induces electron redistribution at surrounding Ni/Fe sites, lowering the formation energy barrier for the *OOH intermediate to 1.60 eV. In situ Raman spectroscopy further reveals that ruthenium single atoms transform nickel active sites into Ni3+-OH species, lowering the activation voltage and significantly accelerating reaction kinetics.
When used as a catalyst in zinc-air batteries, this material demonstrates outstanding comprehensive performance: a maximum power density of 170 mW cm-2 (exceeding the Pt/C + Ir/C system by 42%), a high specific capacity of 783 mAh gZn-1 at a current density of 10 mA cm-2, and stable operation for 350 hours (maintaining 56% charge-discharge efficiency). Crucially, the catalyst retains 93.4% current density after 5000 cycles without structural collapse of its three-dimensional nanosheet architecture. This durability stems from the robust bonding between the in situ-formed NiOOH protective layer and the foam nickel substrate.
This study employs an atomic-level interface regulation strategy, akin to constructing an “atomic-scale electrical distribution network” on the catalyst surface. Ruthenium single atoms serve as electronic distribution hubs, transforming the “bottlenecks” of traditional catalysts into high-speed pathways. This approach establishes a new paradigm for designing highly active, long-lifetime energy conversion catalysts. Its simple synthesis process is compatible with existing battery production lines and has demonstrated potential for large-scale applications.
This work was financially supported by the National Natural Science Foundation of China (22309023, 52301011, and 52231008), the China Postdoctoral Science Foundation (2022M720593), the project of Natural Science Foundation of Chongqing (CSTB2022NSCQ-MSX0270), the youth project of science and technology research program of Chongqing Municipal Education Commission of China (KJQN202501114), and the special funding for research projects of Chongqing Human Resources and Social Security Bureau (2022CQBSHTB1023).
About Nano Research
Nano Research is a peer-reviewed, open access, international and interdisciplinary research journal, sponsored by Tsinghua University and the Chinese Chemical Society, published by Tsinghua University Press on the platform SciOpen. It publishes original high-quality research and significant review articles on all aspects of nanoscience and nanotechnology, ranging from basic aspects of the science of nanoscale materials to practical applications of such materials. After 18 years of development, it has become one of the most influential academic journals in the nano field. Nano Research has published more than 1,000 papers every year from 2022, with its cumulative count surpassing 7,000 articles. In 2024 InCites Journal Citation Reports, its 2024 IF is 9.0 (8.7, 5 years), and it continues to be the Q1 area among the four subject classifications. Nano Research Award, established by Nano Research together with TUP and Springer Nature in 2013, and Nano Research Young Innovators (NR45) Awards, established by Nano Research in 2018, have become international academic awards with global influence.
Article Title
Ru single atoms regulate electron distribution in defective NiFe LDH for enhanced oxygen evolution reaction
Article Publication Date
7-Nov-2025
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of news releases posted to EurekAlert! by contributing institutions or for the use of any information through the EurekAlert system.