Vercammen, J. et al. Shape-selective C–H activation of aromatics to biarylic compounds using molecular palladium in zeolites. Nat. Catal. 3, 1002–1009 (2020).
Lange, J.-P. Performance metrics for sustainable catalysis in industry. Nat. Catal. 4, 186–192 (2021).
Brandi, F., Khalil, I., Antonietti, M. & Al-Naji, M. Continuous-flow production of isosorbide from aqueous-cellulosic derivable feed over sustainable heterogeneous catalysts. ACS Sustain. Chem. Eng. 9, 927–935 (2021).
Dusselier, M., Van Wouwe, P., Dewaele, A., Makshina, E. & Sels, B. F. Lactic acid as a platform chemical in the biobased economy: the role of chemocatalysis. Energy Environ. Sci. 6, 1415–1442 (2013).
te Molder, T. D. J., Kersten, S. R. A., Lange, J.-P. & Ruiz, M. P. From woody biomass to ethylene glycol: inorganics removal boosts the yield. Ind. Eng. Chem. Res. 60, 13515–13522 (2021).
Van Praet, S., Preegel, G., Rammal, F. & Sels, B. F. One-pot consecutive reductive amination synthesis of pharmaceuticals: from biobased glycolaldehyde to hydroxychloroquine. ACS Sustain. Chem. Eng. 10, 6503–6508 (2022).
Schutyser, W. et al. Chemicals from lignin: an interplay of lignocellulose fractionation, depolymerisation, and upgrading. Chem. Soc. Rev. 47, 852–908 (2018).
Sun, Z. et al. Complete lignocellulose conversion with integrated catalyst recycling yielding valuable aromatics and fuels. Nat. Catal. 1, 82–92 (2018).
Liao, Y. et al. A sustainable wood biorefinery for low-carbon footprint chemicals production. Science 367, 1385–1390 (2020).
Dusselier, M., Mascal, M. & Sels, B. F. in Selective Catalysis for Renewable Feedstocks and Chemicals (ed. Nicholas, K.) 1–40 (Springer Cham, 2014).
Lange, J.-P. Towards circular carbo-chemicals – the metamorphosis of petrochemicals. Energy Environ. Sci. 14, 4358–4376 (2021).
Gao, C., Ma, C. & Xu, P. Biotechnological routes based on lactic acid production from biomass. Biotechnol. Adv. 29, 930–939 (2011).
Araji, N. et al. Synthesis of maleic and fumaric acids from furfural in the presence of betaine hydrochloride and hydrogen peroxide. Green Chem. 19, 98–101 (2017).
Yu, Q. et al. A sustainable system for maleic acid synthesis from biomass-derived sugar. J. Chem. Technol. Biotechnol. 95, 751–757 (2020).
Kang, S., Fu, J. & Zhang, G. From lignocellulosic biomass to levulinic acid: a review on acid-catalyzed hydrolysis. Renew. Sustain. Energy Rev. 94, 340–362 (2018).
Xu, W.-P. et al. Conversion of levulinic acid to valuable chemicals: a review. J. Chem. Technol. Biotechnol. 96, 3009–3024 (2021).
Schwartz, T. J., Shanks, B. H. & Dumesic, J. A. Coupling chemical and biological catalysis: a flexible paradigm for producing biobased chemicals. Curr. Opin. Biotechnol. 38, 54–62 (2016).
Shanks, B. H. & Keeling, P. L. Bioprivileged molecules: creating value from biomass. Green Chem. 19, 3177–3185 (2017).
Shanks, B. H. & Broadbelt, L. J. A robust strategy for sustainable organic chemicals utilizing bioprivileged molecules. ChemSusChem 12, 2970–2975 (2019).
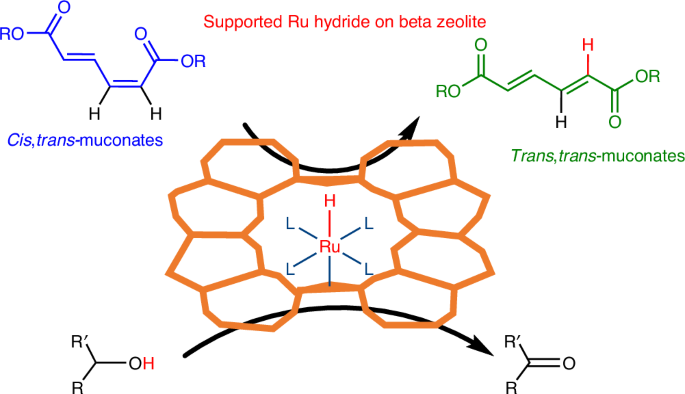
Khalil, I., Quintens, G., Junkers, T. & Dusselier, M. Muconic acid isomers as platform chemicals and monomers in the biobased economy. Green Chem. 22, 1517–1541 (2020).
Briou, B., Améduri, B. & Boutevin, B. Trends in the Diels–Alder reaction in polymer chemistry. Chem. Soc. Rev. 50, 11055–11097 (2021).
Maniar, D. et al. Enzymatic synthesis of muconic acid-based polymers: trans, trans-dimethyl muconate and trans, β-dimethyl hydromuconate. Polymers 13, 2498 (2021).
Zhang, H., Jiang, H., Zhang, Y., Zhang, N. & Xiong, R. Ferroelectric lithography in single‐component organic enantiomorphic ferroelectrics. Angew. Chem. Int. Ed. 61, e202200135 (2022).
Carter, P. et al. Bioenabled platform to access polyamides with built-in target properties. J. Am. Chem. Soc. 22, 9548–9553 (2022).
He, J. et al. Zirconium phosphate supported copper catalyst for selective oxidation of phenol to cis, cis-muconic acid. Appl. Catal. A 664, 119351 (2023).
Klein, B. C. et al. Economics and global warming potential of a commercial-scale delignifying biorefinery based on co-solvent enhanced lignocellulosic fractionation to produce alcohols, sustainable aviation fuels, and co-products from biomass. Energy Environ. Sci. 17, 1202–1215 (2024).
Ling, C. et al. Muconic acid production from glucose and xylose in Pseudomonas putida via evolution and metabolic engineering. Nat. Commun. 13, 4925 (2022).
Shiramizu, M. & Toste, F. D. Expanding the scope of biomass-derived chemicals through tandem reactions based on oxorhenium-catalyzed deoxydehydration. Angew. Chem. Int. Ed. 52, 12905–12909 (2013).
Saraçi, E., Wang, L., Theopold, K. H. & Lobo, R. F. Bioderived muconates by cross‐metathesis and their conversion into terephthalates. ChemSusChem 11, 773–780 (2018).
Quintens, G., Vrijsen, J., Adriaensens, P., Vanderzande, D. & Junkers, T. Muconic acid esters as bio-based acrylate mimics. Polym. Chem. 10, 5555–5563 (2019).
Carraher, J. M., Pfennig, T., Rao, R. G., Shanks, B. H. & Tessonnier, J. P. cis,cis-Muconic acid isomerization and catalytic conversion to biobased cyclic-C6-1,4-diacid monomers. Green Chem. 19, 3042–3050 (2017).
Lu, R. et al. Production of diethyl terephthalate from biomass-derived muconic acid. Angew. Chem. Int. Ed. 55, 249–253 (2016).
Rammal, F., Gaumont, A. C. & Lakhdar, S. Metal-free visible-light-mediated aromatization of 1,2-dihydronaphthalenes. Eur. J. Org. Chem. 2020, 1482–1485 (2020).
Carraher, J. M. et al. Solvent-driven isomerization of cis,cis-muconic acid for the production of specialty and performance-advantaged cyclic biobased monomers. Green Chem. 22, 6444–6454 (2020).
Gopalakrishnan, D. K., Bhardwaj, S., Kumar, S., Karmakar, T. & Vaitla, J. Carbene-mediated stereoselective olefination of vinyl sulfoxonium ylides with diazo compounds and acetals. Chem. Commun. 60, 3846–3849 (2024).
Grundmann, C. Zur kenntnis der oxydation von phenolen mit peressigsäure. Ber. Dtsch. Chem. Ges. 69, 1755–1757 (1936).
Settle, A. E. et al. Iodine-catalyzed Isomerization of dimethyl muconate. ChemSusChem 11, 1768–1780 (2018).
Frost, J. W., Miermont, A., Schweitzer, D. & Bui, V. Preparation of trans, trans muconic acid and trans, trans muconates. US patent US0314243 A1 (2010).
Bui, V., MacRae, D. & Schweitzer, D. Methods for producing isomers of muconic acid and muconate salts. US patent US0030215 (2013).
Tessonnier, J. P., Carraher, J. M., Pfennig, T. & Shanks, B. Isomerization of muconic acid. US patent US9957218 B2 (2018).
Khalil, I. et al. Solvent-driven isomerization of muconates in DMSO: reaction mechanism and process sustainability. Green Chem. 26, 5852–5861 (2024).
Peeters, E. et al. Tandem reduction–reoxidation augments the catalytic activity of Sn-beta zeolites by redispersion and respeciation of SnO2 clusters. Chem. Mater. 33, 9366–9381 (2021).
Kerstens, D. et al. Fast and selective solvent-free branching of unsaturated fatty acids with hierarchical ZSM-5. ACS Sustain. Chem. Eng. 9, 4357–4362 (2021).
Shah, M. A. et al. Catalytic amination of lactic acid using Ru–zeolites. Dalton Trans. 51, 10773–10778 (2022).
Philippaerts, A. et al. Design of Ru–zeolites for hydrogen-free production of conjugated linoleic acids. ChemSusChem 4, 757–767 (2011).
Yue, C. J., Liu, Y. & He, R. Olefins isomerization by hydride-complexes of ruthenium. J. Mol. Catal. Chem. 259, 17–23 (2006).
Wang, A., Li, J. & Zhang, T. Heterogeneous single-atom catalysis. Nat. Rev. Chem. 2, 65–81 (2018).
Ge, L. et al. Synergistic catalysis of Ru single-atoms and zeolite boosts high-efficiency hydrogen storage. Appl. Catal. B 319, 121958 (2022).
Matthiesen, J. E., Carraher, J. M., Vasiliu, M., Dixon, D. A. & Tessonnier, J. P. Electrochemical conversion of muconic acid to biobased diacid monomers. ACS Sustain. Chem. Eng. 4, 3575–3585 (2016).
Qiu, J.-Z. et al. Pure siliceous zeolite-supported Ru single-atom active sites for ammonia synthesis. Chem. Mater. 31, 9413–9421 (2019).
Tekin, K., Hao, N., Karagoz, S. & Ragauskas, A. J. Ethanol: a promising green solvent for the deconstruction of lignocellulose. ChemSusChem 11, 3559–3575 (2018).
Yang, H. et al. Isolated single‐atom ruthenium anchored on beta zeolite as an efficient heterogeneous catalyst for styrene epoxidation. ChemNanoMat 6, 1647–1651 (2020).
Sun, R. et al. Heterogeneous catalysts for CO2 hydrogenation to formic acid/formate: from nanoscale to single atom. Energy Environ. Sci. 14, 1247–1285 (2021).
Ghoreishian, S. M., Shariati, K., Huh, Y. S. & Lauterbach, J. Recent advances in ammonia synthesis over ruthenium single-atom-embedded catalysts: a focused review. Chem. Eng. J. 467, 143533 (2023).
Mon, M. et al. Stabilized Ru[(H2O)6]3+ in confined spaces (MOFs and zeolites) catalyzes the imination of primary alcohols under atmospheric conditions with wide scope. ACS Catal. 8, 10401–10406 (2018).
Wang, D. & Astruc, D. The golden age of transfer hydrogenation. Chem. Rev. 115, 6621–6686 (2015).
Capeletti, M. R., Balzano, L., de la Puente, G., Laborde, M. & Sedran, U. Synthesis of acetal (1,1-diethoxyethane) from ethanol and acetaldehyde over acidic catalysts. Appl. Catal. A 198, L1–L4 (2000).
Hao, W. et al. Ru-catalyzed enantioselective hydrogenation of 2-pyridyl-substituted alkenes and substrate-mediated H/D exchange. ACS Catal. 12, 1150–1160 (2022).
Kita, Y., Kuwabara, M., Yamadera, S., Kamata, K. & Hara, M. Effects of ruthenium hydride species on primary amine synthesis by direct amination of alcohols over a heterogeneous Ru catalyst. Chem. Sci. 11, 9884–9890 (2020).
Wachs, I. E. & Bañares, M. A. (eds) Springer Handbook of Advanced Catalyst Characterization (Springer Cham, 2023).
Wang, X. & Andrews, L. Infrared spectra and theoretical calculations for Fe, Ru, and Os metal hydrides and dihydrogen complexes. J. Phys. Chem. A 113, 551–563 (2009).
Settle, A. E. et al. Heterogeneous Diels–Alder catalysis for biomass-derived aromatic compounds. Green Chem. 19, 3468–3492 (2017).
Shang, Y., Xu, X., Gao, B., Wang, S. & Duan, X. Single-atom catalysis in advanced oxidation processes for environmental remediation. Chem. Soc. Rev. 50, 5281–5322 (2021).
Devos, J. et al. Engineering low-temperature ozone activation of zeolites: process specifics, possible mechanisms and hybrid activation methods. Chem. Eng. J. 431, 133862 (2022).
Zavala‐Sanchez, L., Khalil, I., Oliviero, L., Paul, J. & Maugé, F. Structure and quantification of edge sites of WS2/Al2O3 catalysts coupling IR/CO spectroscopy and DFT calculations. ChemCatChem 12, 2066–2076 (2020).
Emeis, C. A. Determination of integrated molar extinction coefficients for infrared absorption bands of pyridine adsorbed on solid acid catalysts. J. Catal. 141, 347–354 (1993).
Devos, J., Robijns, S., Van Goethem, C., Khalil, I. & Dusselier, M. Interzeolite conversion and the role of aluminum: toward generic principles of acid site genesis and distributions in ZSM-5 and SSZ-13. Chem. Mater. 33, 2516–2531 (2021).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
te Velde, G. et al. Chemistry with ADF. J. Comput. Chem. 22, 931–967 (2001).
Martini, A. et al. PyFitIt: the software for quantitative analysis of XANES spectra using machine-learning algorithms. Comput. Phys. Commun. 250, 107064 (2020).