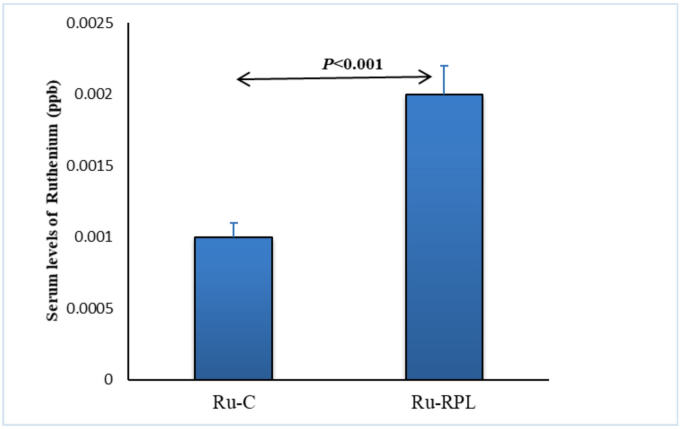
The present study reveals a clear association between elevated serum Ru levels, OS, and DNA damage in women with RPL. Compared with healthy controls, the RPL group exhibited significantly higher MDA and 8-OHdG levels, along with reduced GSH concentrations, indicating a pronounced disruption of redox homeostasis and enhanced DNA damage. Serum Ru correlated positively with MDA and 8-OHdG and negatively with GSH, indicating that higher Ru levels are associated with greater oxidative imbalance and DNA damage in women with RPL.
Environmental pollutants, arising from human activity as well as natural sources, are ubiquitous in air, water, soil, and food. Such pollutants can adversely affect pregnancy, leading to outcomes ranging from polycystic ovarian syndrome, embryonic loss, spontaneous abortion to intrauterine growth restriction, and neurodevelopmental disorders18,19,20,21,22. Among these pollutants, rare earth elements (REEs) have drawn attention to their potential reproductive toxicity. Ru has expanding industrial and technological applications and is being explored for therapeutic uses such as anticancer agents. Yet, its impact on human reproductive health remains poorly defined, with no established safety thresholds and only limited preclinical data. To date, however, no clinical studies have directly examined circulating Ru levels in women with RPL.
To my knowledge, this represents the first clinical investigation assessing circulating Ru levels in women with RPL and their association with OS and DNA damage, thereby filling a critical gap in human data and complementing existing preclinical and animal studies on Ru reproductive toxicity. Compared with healthy controls, women with RPL exhibited significantly higher serum Ru concentrations, increased MDA and 8-OHdG levels, and decreased GSH. Ru correlated positively with MDA (r = 0.30) and 8-OHdG (r = 0.47), and negatively with GSH (r = − 0.80), indicating a strong inverse relationship with antioxidant capacity. Multivariate regression adjusting for age, BMI, and parity showed that Ru independently predicted higher MDA (β = +0.18, adjusted R² = 0.38) and 8-OHdG (β = +0.72, adjusted R² = 0.46), and lower GSH (β = − 0.60, adjusted R² = 0.63), collectively explaining 38–63% of the variance in these biomarkers. Interestingly, women with RPL in this cohort had significantly lower BMI compared with controls. While obesity is often regarded as a miscarriage risk factor, this observation supports prior evidence that both low and high BMI may impair reproductive outcomes23. Importantly, adjusting for BMI did not alter the associations between Ru, OS markers, and DNA damage, suggesting that these relationships are not driven by body weight differences. An additional consideration is the timing of sample collection. Samples from women with RPL were collected shortly after miscarriage. Miscarriage itself can contribute to oxidative alterations, including elevated MDA and 8-OHdG and reduced GSH, due to acute inflammatory and stress responses. Therefore, while miscarriage may contribute to oxidative alterations, capturing this post-miscarriage profile provides valuable insight into the redox environment in which pregnancy was lost. Additionally, although BMI influences redox balance, regression analyses adjusting for BMI confirmed that the associations between Ru, OS markers, and DNA damage remained significant, supporting the robustness of the findings.
Experimental evidence indicates that certain Ru compounds can act as pro-oxidants, impair redox balance, and induce DNA damage19. Placental development naturally involves controlled OS, which supports normal placentation. However, excessive OS can disrupt pregnancy by damaging lipids, proteins, and DNA. MDA, a product of lipid peroxidation, compromises membrane structure and enzyme function and is consistently elevated in women with RPL24,25,26,27. In parallel, depletion of the key antioxidant GSH reflects impaired redox homeostasis, as reported in multiple independent studies. Chen et al.28 linked YAP1-mediated trophoblast ferroptosis to RPL via lipid peroxidation and MDA generation30. El-Sikaily et al.., 2023 documented increased lipid peroxidation and reduced antioxidant defenses in RPL women29. Al-Sheikh et al..,2019 reported disturbances in key oxidant and antioxidant molecules among Saudi women with recurrent miscarriage30. More recently, Tabassum et al.., 2025 reported decreased antioxidant capacity in RPL women22. Collectively, these observations underscore oxidative imbalance as a central mechanism connecting environmental or molecular insults to pregnancy loss.
Beyond lipid peroxidation, OS can directly compromise DNA integrity, generating lesions such as apurinic sites and oxidized nucleobases16,31,32. Among these, 8-OHdG is a well-established marker of oxidative DNA damage and provides insight into the genomic instability associated with pregnancy loss33,34. The study findings show significantly higher 8-OHdG in RPL women, correlating positively with serum Ru levels. Excessive OS can induce double strand breaks in gametes, thereby increasing the risk of RPL. Elevated 8-OHdG levels have been reported in placental tissue from unexplained fetal deaths and in the sera of women with pregnancy loss31,35.
Experimental studies demonstrate that Ru compounds can induce mitochondrial dysfunction, DNA strand breaks, impaired gametogenesis, and embryotoxicity, although toxicity varies by chemical species8,36,37. Beyond Ru, other REEs also influence redox balance and DNA stability. Direct epidemiological data on circulating Ru concentrations in human cohorts are scarce, limiting quantitative comparisons with the present findings. Most biomonitoring studies do not include either Ru or report levels below detection thresholds. However, several epidemiological investigations have linked exposure to other rare earth elements (REEs) with adverse reproductive outcomes. While these reports do not provide direct Ru comparisons, they support the broader plausibility that REE exposure may influence reproductive health, reinforcing the relevance of present observations. Wei et al.., 2017 reported an association between maternal hair REE levels and an increased risk of neural tube defects, suggesting that maternal exposure to REEs can have teratogenic effects during early pregnancy38. Similarly, Li et al.., 2021 found that light REE exposure in women undergoing IVF–embryo transfer was linked with poorer reproductive outcomes, highlighting potential impacts on implantation or early embryonic development39. Additionally, Liu et al.., 2021 observed that REE concentrations in umbilical cord blood were associated with a higher risk of orofacial clefts, indicating that fetal accumulation of these metals may influence developmental processes40. Together, these data support the biological plausibility that environmental or dietary Ru exposure could contributes to reproductive dysfunction20,22,31. Collectively, the findings indicate that elevated circulating Ru is strongly associated with OS and DNA damage in women with RPL, including a pronounced inverse relationship with antioxidant capacity (GSH). Although causality cannot be inferred from this observational study, the consistent associations with established biomarkers and supporting preclinical evidence highlight Ru as a potential environmental risk factor for reproductive complications. Ruthenium, while attracting interest for its potential anticancer properties, may pose reproductive concerns due to its association with OS and DNA damage4. Pregnant women exposed to Ru, whether therapeutically or environmentally, may face an elevated risk of miscarriage, impaired fetal growth, and developmental abnormalities, particularly during early gestation. Therapeutic applications henceforth require targeted delivery, precise dosing, and careful monitoring, whereas incidental environmental exposure should be minimized. Future research involving larger cohorts and longitudinal study designs is warranted to clarify causal relationships, assess strategies to reduce Ru exposure or enhance antioxidant defenses, and investigate interactions with DNA repair and antioxidant systems
Limitations
This study has limitations. The sample size was relatively small, and the stricter RPL definition used focusing on women with a more severe and consistent history reduces heterogeneity but may limit generalizability. Participants with comorbidities or occupational metal exposure were excluded, and key potential confounders such as diet, supplement use (including prenatal vitamins and antioxidants), lifestyle factors, and environmental exposures were not recorded. Blood samples from RPL women were collected shortly after miscarriage, and both miscarriage itself and BMI differences between groups may independently affect OS markers. Although all multivariate regression analyses were adjusted for BMI, residual confounding related to body composition cannot be completely excluded. Specific sources of Ru exposure were not assessed, preventing direct linkage of circulating levels to environmental or lifestyle factors. Despite these limitations, the findings provide preliminary evidence linking elevated Ru to OS and DNA damage in women with RPL.